Company Profile
Biozen, LLC is a US-based digital health company and medical device manufacturer, headquartered in Oklahoma City, Oklahoma, USA, with research and development in Atlanta, Georgia. Founded in 2010, the company operates as an independent privately-held entity employing approximately 26 people, serving clinicians, healthcare partners and adult patients across the United States.
Core Products & Technologies
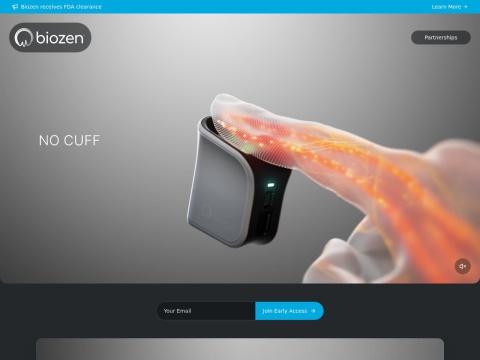
Cuffless Blood Pressure Monitoring
• Biozen BP1000: FDA-cleared ultra-compact fingertip blood pressure monitor delivering calibration-free spot measurements for adults ages 22-59
• Companion Mobile App: Visual-guided interface that logs readings and maintains comprehensive measurement history
Platform Technology
• Proprietary Pressure Sensing: Direct arterial occlusion detection through fingertip application
• Photoplethysmography (PPG): Optical blood volume change tracking paired with pressure signals
• Physiologically Informed Algorithms: Oscillometric-method computation without per-user calibration against arm cuffs
Market Position & Certifications
Biozen holds a pioneering position in the cuffless blood pressure monitoring market, competing with Biobeat and Aktiia. Key strengths include:
• 15+ years of biosensor and digital health research and development heritage
• Direct Measurement Principle: Absolute blood pressure measurement from the fingertip without estimation or correlation models
• Regulatory compliance: FDA 510(k) cleared (May 2026), validated against ISO 81060-2:2018 for equivalence to cuff-based references
• Intellectual property: Platform protected by more than 68 granted US and international patents
Corporate Timeline
2010 — Founded as a digital health innovator focused on advanced biosensors
2023 — Completed later-stage venture capital financing to scale commercial operations
2026 — Named finalist in ACC Innovation Pitch Challenge; presented prospective clinical validation data at ACC
2026 — Received FDA 510(k) clearance for BP1000 on May 11; opened early access program for clinicians and partners
Target Markets & Applications
• Home Blood Pressure Monitoring: BP1000 for adults seeking convenient, cuffless spot measurements in home environments
• Primary Care & Cardiology Clinics: Calibration-free clinical-grade readings to improve patient compliance and monitoring frequency
• Future Device Integration: BP Sensor platform licensing for smartphones, wearables and connected health ecosystems
Contact Information
Global Headquarters
Address: 7023 N. Classen Blvd., Oklahoma City, Oklahoma 73116, USA
Phone: +1 405-413-5201
Email: aw@biozen.com
Website: www.biozen.com
Research & Development Center
Address: Atlanta, Georgia, USA