Company Profile
Life Spine, Inc. is a US-based manufacturer of spinal implants and minimally invasive surgical technologies, headquartered in Huntley, Illinois. Founded in 2004 by Michael Butler in Indianapolis, the privately held company relocated to the northwest suburbs of Chicago and now employs over 70 people worldwide. Life Spine serves orthopedic and neurosurgery communities in over 30 countries, supported by a global intellectual property portfolio of 180+ patents and 220+ filed patent applications.
Core Products & Technologies
Expandable & Interbody Systems
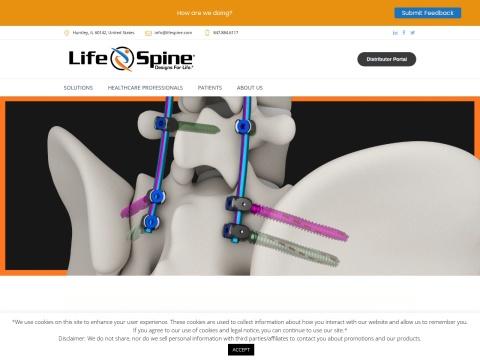
• ProLift Expandable Spacer System: In-situ adjustable interbody fusion device; line includes ProLift, ProLift Micro, and ProLift Lateral for TLIF, PLIF, and LLIF approaches
• ProLift Pivot Expandable Spacer System: Full articulation with in-situ expansion up to 5mm, bulleted tip, and OSSEO-LOC surface technology; FDA cleared January 2025
• PLATEAU-X Ti Lateral Lumbar Spacer: Titanium lateral interbody device with self-distracting leading edge and convex endplate contact design
Cervical & Thoracolumbar Fixation
• GRUVE+ Cervical Plating System: Anterior cervical plate with sub-flush design, CAM locking mechanism, and compatibility with PLATEAU-C and Pro-Link platforms; FDA cleared February 2025
• ARx SAI Spinal Fixation System: Posterior fixation with sacral alar iliac trajectory, self-drilling / self-harvesting screws, and low-profile cobalt chrome tulip head; expanded FDA clearance April 2026 for 5.5mm and 6.0mm titanium and cobalt chrome rods
• NAUTILUS Thoracolumbar System: Comprehensive posterior fixation with double-lead thread geometry and generous screw angulation
SI Joint Fusion & Accessory Technologies
• SImpact SI Screw: Self-drilling, self-harvesting sacroiliac fixation screw with autograft recruitment design
• OSSEO-LOC Surface Technology: Proprietary surface treatment applied to titanium implants to promote osseointegration and long-term stability
Market Position & Certifications
Life Spine holds a specialized position in the minimally invasive spinal implant segment, competing with Medtronic, Stryker, DePuy Synthes (Johnson & Johnson), NuVasive, Globus Medical, and Zimmer Biomet. Key strengths include:
• 20+ years of spinal innovation heritage
• Micro Invasive™ technology: Proprietary expandable implants designed to reduce operating time, cost, and postoperative complications
• Regulatory compliance: FDA 510(k) cleared, CE marked; ProLift portfolio confirmed MR Conditional per ASTM standards (February 2026)
• Surgeon-driven design: Instruments streamlined for procedural efficiency and reproducibility
Corporate Timeline
2004 — Founded by Michael Butler in Indianapolis, Indiana
2014 — Relocated headquarters to Huntley, Illinois
2025 — Richard Mueller appointed President & CEO; ProLift Pivot and GRUVE+ receive FDA 510(k) clearance
2026 — ProLift expandable portfolio confirmed MR Conditional; ARx SAI receives expanded indications FDA clearance
Target Markets & Applications
• Degenerative Conditions: Disc disease, spinal stenosis, and spondylolisthesis fusion
• Spinal Trauma & Deformity: Fracture stabilization, scoliosis, and kyphosis correction
• Minimally Invasive Fusion: TLIF, PLIF, LLIF, and endoscopic interbody fusion procedures
• SI Joint Dysfunction: Sacroiliac joint stabilization and fixation
Contact Information
Global Headquarters
Address: 13951 S Quality Drive, Huntley, Illinois 60142, USA
Phone: (847) 884-6117
Email: info@lifespine.com
Website: www.lifespine.com