Company Profile
Tandem Diabetes Care, Inc. is a US-based manufacturer of insulin delivery systems, headquartered in San Diego, California, United States. Founded in 2006 as Phluid, Inc. and reincorporated as Tandem Diabetes Care in 2008, the company operates as an independent publicly traded entity on NASDAQ (TNDM), serving insulin-dependent patients in over 50 countries.
Core Products & Technologies
Insulin Pumps
• t:slim X2 Insulin Pump: A touchscreen insulin pump with 300-unit capacity, integrated color display, and remote software update capability
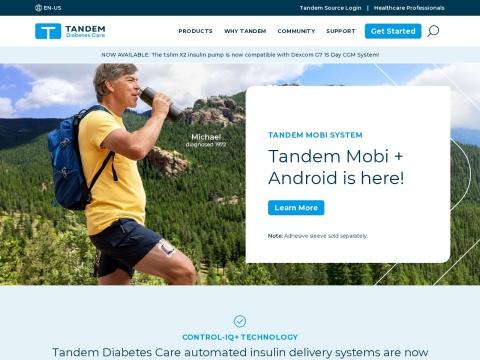
• Tandem Mobi System: The world's smallest durable automated insulin delivery system, smartphone-controlled via iOS and Android, with 200-unit capacity and wireless charging
Automated Insulin Delivery Technology
• Control-IQ+ Technology: An advanced hybrid closed-loop algorithm that automatically adjusts basal insulin and delivers correction boluses based on CGM readings
• t:connect Mobile App: A cloud-based diabetes management platform enabling remote monitoring and mobile bolus delivery
CGM Compatibility
• Dexcom G6 / G7: Integrated continuous glucose monitoring for real-time automated insulin adjustments
• Abbott FreeStyle Libre 2+ / 3+: Expanded sensor compatibility for broader patient choice
Market Position & Certifications
Tandem Diabetes Care holds a top-three position in the global insulin pump market, competing with Medtronic and Insulet. Key strengths include:
• 20+ years of US engineering heritage in diabetes device innovation
• Control-IQ+ Technology: FDA-cleared for Type 1 (ages 2+) and Type 2 diabetes (ages 18+)
• Regulatory compliance: FDA cleared, Health Canada licensed
• Global installed base: Nearly 500,000 users worldwide as of 2024
• Software-updatable platform: Remote feature upgrades without hardware replacement
Corporate Timeline
2006 — Founded as Phluid, Inc. in San Diego, California
2008 — Reincorporated as Tandem Diabetes Care, Inc.
2012 — FDA clearance for the t:slim Insulin Pump, the first touchscreen insulin pump
2013 — Initial Public Offering on the Nasdaq stock exchange (TNDM)
2019 — FDA clearance for Control-IQ advanced hybrid closed-loop technology
2023 — FDA clearance for Tandem Mobi, the world's smallest durable AID system
2025 — FDA clearance for Control-IQ+ for Type 2 diabetes
2026 — FDA clearance and full commercial launch of Mobi Android compatibility in the United States
Target Markets & Applications
• Type 1 Diabetes Management: t:slim X2 and Tandem Mobi with Control-IQ+ for automated insulin delivery in patients aged 2 and older
• Type 2 Diabetes Therapy: Control-IQ+ expanded indication for insulin-requiring Type 2 patients aged 18 and older
• Pediatric Care: AID systems indicated for children and adolescents with insulin-dependent diabetes
Contact Information
Global Headquarters
Address: 12400 High Bluff Drive, San Diego, California 92130, United States
Investor Relations
Stock: NASDAQ: TNDM
Website: tandemdiabetes.com
Investor Relations: investor.tandemdiabetes.com